HKUST Team Unveils Innovative Technology for Precise Reading and Control of Brain Circuits to Advance Brain Disease Research
A cross-disciplinary team led by Prof. QU Jianan from the Department of Electronic and Computer Engineering and Prof. Julie L. SEMMELHACK from the Division of Life Science at The Hong Kong University of Science and Technology (HKUST) has developed a powerful laser control technique. Functioning as a “smart dimmer”, it can selectively adjust the brightness of every pixel during laser scanning, and prevent unintended neural activation, thereby significantly enhancing the precision of all-optical brain manipulation and imaging. This innovation will drive research into brain disease mechanisms and facilitate small animal disease models for new drug development. The finding results, titled “Active pixel power control for crosstalk-free all-optical neural interrogation” have been published in the prestigious international journal Nature Communications.
Address crosstalk problem in all-optical interrogation of the active brain
In recent years, advancements in “all-optical interrogation” of neural circuits have made it possible to pinpoint exactly which neurons trigger the corresponding movements, perceptions, or emotional responses. This achievement is enabled by two scientific breakthroughs. First, genetically encoded activity sensors, such as calcium indicators, cause neurons to glow and become visible under modern microscopes when they fire. Second, optogenetic actuators, the light-sensitive proteins like channelrhodopsins, allow specific neurons to turn on or off using flashes of light.
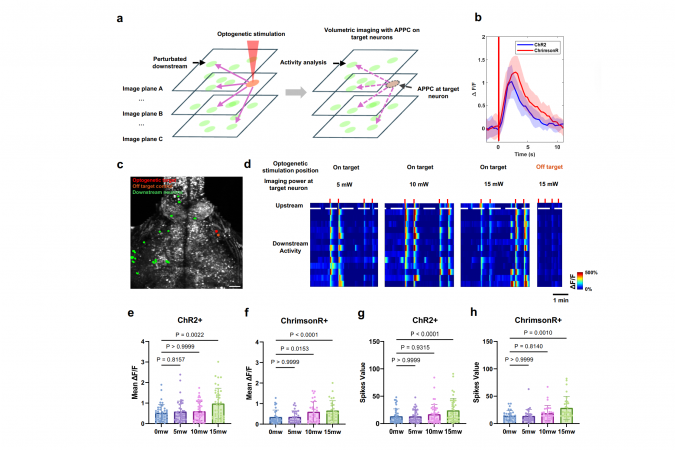
While this approach offers advantages of high speed and single-cell precision, a key challenge persists: the infrared laser, intended for passive observation, can itself drive other neurons to fire, making it difficult to distinguish natural brain activity from experimental artifacts. This phenomenon, known as “crosstalk,” affects accuracy.
“All-optical approaches hold great promise, but crosstalk has been a major barrier,” said Prof. Qu. “When you use microscopic imaging to observe neural circuits, the imaging light secretly pushes them around, influencing the operation of neurons and even the entire neural circuit.”
Smart dimmer for every pixel
To preclude such unwanted artificial intervention, HKUST team has developed the “Active Pixel Power Control (APPC)” strategy, which functions like a smart, real-time dimmer switch. Guided by custom mapping software that identifies where optogenetic proteins are expressed, a fast acousto-optic modulator dynamically adjusts laser power of each scanning pixel. It delivers reduced or even zero power to specific neurons with varying levels of optogenetic protein expression while maintaining uniform and stable light intensity across other brain regions, thereby reducing the issue of crosstalk.
“APPC lets us protect the very neurons we want to stimulate, without sacrificing recording quality elsewhere. It is a simple yet technically challenging idea,” explained Prof. Semmelhack.
Larvae zebrafish, a small, transparent vertebrate widely used in brain research, shares over 70% genetic similarity with the human brain. To test the capabilities of this technology, the team applied it to the larval zebrafish. In an in vivo study of the zebrafish brain, APPC successfully preserved signal quality of neuronal signals while suppressing optogenetic artifacts and reducing the issue of crosstalk.
An important strength of APPC is its compatibility with standard two-photon microscopes extensively used worldwide, enabling practical and cost-effective implementation without requiring complete system replacement. Beyond this demonstration, APPC strategy can readily extend to other animal models, particularly mice, the most commonly used model in modern neuroscience.
Prof. Qu elaborated, “All-optical interrogation enables researchers to precisely investigate how brain circuits drive behavior under natural physiological conditions. Now, APPC brings that vision closer by solving the crosstalk issue, one of the biggest technical roadblocks.”
Prof. Semmelhack added, “This study not only demonstrates how precision optics can be integrated with in vivo circuit neuroscience, but also exemplifies what happens when engineering and biology teams collaborate. We are excited to see how other groups will utilize APPC to further explore brain mechanisms and pathology.”
Prof. Qu and Prof. Semmelhack are the co-corresponding authors of this research. Mr. YAN Gewei, PhD student of the Department of Electronic and Computer Engineering, and Dr. TIAN Guangnan, postdoctoral fellow of the Division of Life Science, are the co-first authors. The project is supported by the Collaborative Research Fund and the General Research Fund under the Research Grants Council in Hong Kong.
(This news was originally published by the HKUST Global Engagement and Communications Office here.)